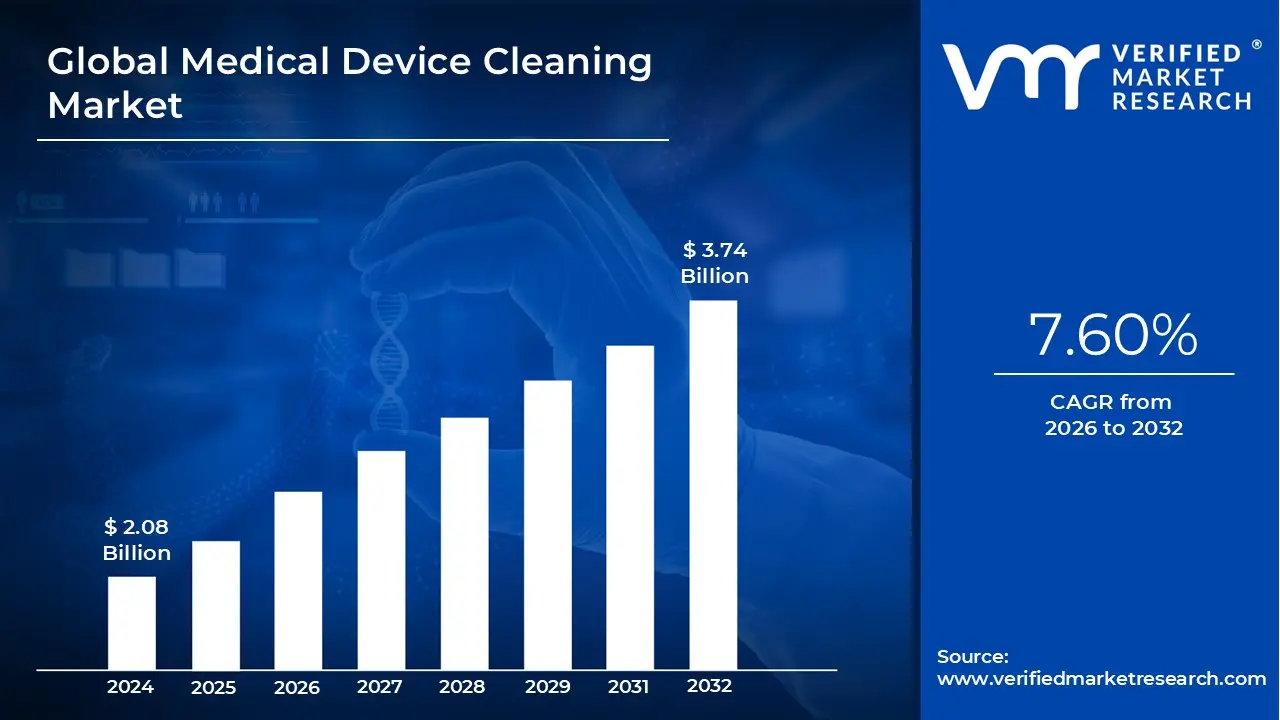
Lewes, Delaware, Feb. 27, 2026 (GLOBE NEWSWIRE) -- Verified Market Research® indicates that the Global Medical Device Cleaning Market was valued at USD 2.08 Billion in 2024 and is projected to reach USD 3.74 Billion by 2032, expanding at a CAGR of 7.60% during the forecast period of 2026–2032. The growth trajectory reflects intensifying regulatory scrutiny, growing surgical interventions, and technological modernization within sterile processing departments.
Download a free sample to access exclusive Insights, Data Charts, And Forecasts From The Medical Device Cleaning Sample Report
Explore the full Medical Device Cleaning Market
TOC: 202 Pages
Tables: 118
Figures: 97
Medical Device Cleaning Market Overview
Market Drivers
1. Escalating Infection Prevention and Compliance Requirements
Healthcare systems worldwide are operating under increasingly stringent infection control frameworks, compelling hospitals, ambulatory surgical centers, and specialty clinics to modernize device reprocessing practices. Medical device cleaning, positioned as a critical precursor to sterilization and high-level disinfection, is gaining strategic visibility among quality assurance and procurement teams. For enterprise buyers, investments in validated cleaning systems mitigate cross-contamination risks, reduce liability exposure, and align operations with accreditation standards, thereby reinforcing long-term procurement strategies.
2. Expansion of Reusable Medical Devices and Surgical Throughput
The growing utilization of reusable surgical instruments, endoscopes, and minimally invasive devices is significantly amplifying cleaning and decontamination volumes. Facilities are optimizing sterile processing workflows to handle higher instrument turnover without compromising efficacy. This demand dynamic creates measurable opportunities for automated washer-disinfectors, enzymatic detergents, and monitoring solutions. Market research stakeholders recognize this as a scalable growth lever, as device longevity initiatives and sustainability targets drive preference for advanced cleaning technologies across large healthcare networks.
3. Technological Advancements in Automated Reprocessing Systems
Innovation in automated cleaning platforms, ultrasonic systems, and digital validation tools is transforming reprocessing reliability and labor efficiency. Smart systems equipped with cycle monitoring, data logging, and compliance tracking are enabling traceability and audit readiness. From a strategic procurement perspective, these technologies offer quantifiable operational savings and regulatory safeguards. Investors and enterprise buyers view automation as a defensible competitive differentiator, particularly in high-throughput environments where consistency and documentation are mission-critical.
Download a free sample to access exclusive Insights, Data Charts, And Forecasts From The Medical Device Cleaning Sample Report.
Market Restraints
1. High Capital and Operational Cost Structures
The acquisition and maintenance of automated cleaning systems involve considerable capital expenditure, particularly for mid-sized and resource-constrained facilities. Budgetary constraints often delay modernization initiatives, influencing uneven market penetration. Cost sensitivities are especially pronounced in emerging healthcare markets, where procurement decisions are tightly governed by reimbursement and funding limitations.
2. Complex Regulatory and Validation Obligations
Medical device cleaning processes are governed by intricate regulatory standards and validation protocols, requiring specialized expertise and continuous monitoring. Compliance burdens may discourage smaller providers from adopting advanced systems. Regional disparities in regulatory frameworks further complicate multinational deployment strategies and vendor alignment.
3. Workforce Training and Process Variability Risks
Effective device cleaning depends heavily on staff competency, standardized protocols, and adherence to manufacturer guidelines. Variability in training and operational discipline can compromise outcomes, generating reputational and safety risks. Healthcare organizations must allocate sustained resources to education and quality control, impacting total cost of ownership calculations.
Medical Device Cleaning Market Geographical Dominance
North America maintains a dominant position within the medical device cleaning market, supported by mature healthcare infrastructure, robust regulatory enforcement, and widespread automation across sterile processing departments. The United States leads adoption due to high surgical volumes and rigorous accreditation requirements, while Canada demonstrates steady modernization initiatives. Europe follows closely, with Germany, the United Kingdom, and France emphasizing compliance-driven procurement and sustainable reprocessing practices. Asia-Pacific represents a high-growth region, fueled by expanding hospital capacity, rising procedural demand, and regulatory evolution in countries such as China, Japan, and India. Emerging markets across Latin America and the Middle East are progressively investing in infection control technologies, signaling long-term opportunity.
Medical Device Cleaning Market Key Players
Competitive intelligence remains central to strategic decision-making, as vendors differentiate through automation capabilities, detergent chemistries, validation tools, and service models. Prominent participants in the global medical device cleaning market include 3M Company, Advanced Sterilization Products, Belimed AG, Cantel Medical Corporation, Cardinal Health, Ecolab, Inc., EMS GmbH, Getinge Group, Johnson & Johnson, Medtronic plc, Mölnlycke Health Care AB, Olympus Corporation, PerkinElmer, Inc., Reprocessing Services Ltd, Stryker Corporation.
Medical Device Cleaning Market Segmentation
The Medical Device Cleaning Market is analyzed across the following dimensions:
By Product
- Detergents
- Chemicals
By Application
- Surgical Instruments
- Ultrasound Probes
- Endoscopes
- Dental Instruments
- Others
By End User
- Hospitals & Clinics
- Dental Clinics & Hospitals
- Diagnostic Centers
- Others
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
To gain complete access with Corporate Or Enterprise Licensing, Visit The Medical Device Cleaning Market.
Medical Device Cleaning Market Strategic Insights
Market attractiveness varies by region, with developed markets favoring automation upgrades and emerging regions emphasizing capacity expansion. Entry barriers remain moderate to high due to regulatory compliance, validation expertise, and capital intensity. Pricing strategies increasingly reflect value-based procurement, where lifecycle cost efficiencies outweigh upfront expenditure. Partnerships with healthcare institutions, compliance bodies, and service providers are shaping vendor positioning. For investors and enterprise stakeholders, scalable automation portfolios and regulatory alignment represent critical success factors.
Key Highlights of the Report
- Comprehensive market size and forecast analysis
- Detailed segmentation and technology assessment
- Regional demand and investment patterns
- Competitive landscape and vendor benchmarking
- Evaluation of drivers, challenges, and risk factors
Why This Report Matters?
This report equips decision-makers with structured intelligence to evaluate growth opportunities, technology investments, and competitive positioning. It supports revenue planning, procurement strategies, and market entry assessments through data-driven insights and forward-looking analysis.
Who Should Read This Report?
- Market research and consulting professionals
- Medical device and reprocessing technology manufacturers
- Enterprise buyers and procurement strategists
- Investors and corporate planners
Related Reports
Global Medical Plastics Market Size By Product Type (Polyethylene (PE), Polypropylene (PP), Polycarbonate (PC)), By Process Technology (Extrusion, Injection Molding, Blow Molding), By Application (Medical Device Packaging, Medical Components, Orthopedic Implant Packaging, Orthopedic Soft Goods), By Geographic Scope And Forecast
Global Pharmaceutical 3PL Service Market Size By Service Type (Transportation & Freight Services, Warehousing & Distribution, Inventory Management, Order Fulfillment, Cold Chain Logistics, Packaging and Repackaging, Value Added Services), By End-User (Pharmaceutical Manufacturers, Biotechnology Firms, Medical Device Companies, Contract Research Organizations (CROs), Retail Pharmacies, E Pharmacies), By Geographic Scope and Forecast
Global Disposable Medical Devices Sensors Market Size By Product Type (Temperature Sensors, Pressure Sensors), By Application (Patient Monitoring, Diagnostics), By End-User (Hospitals, Home Care), By Geographic Scope And Forecast
Global Medical Device Packaging Market Size By Product (Pouches And Bags, Trays, Boxes, Clamshells), By Material (Plastic, Paper And Paperboard, Metal), By Application (Equipment And Tools, Devices, IVD, Implants), By Geographic Scope And Forecast
Top 7 Medical Device Cleaning Companies maintaining quality and safety together
Visualize Medical Device Cleaning Market using Verified Market Intelligence -:
Verified Market Intelligence is our BI Enabled Platform for narrative storytelling in this market. VMI offers in-depth forecasted trends and accurate Insights on over 20,000+ emerging & niche markets, helping you make critical revenue-impacting decisions for a brilliant future.
VMI provides a holistic overview and global competitive landscape with respect to Region, Country, Segment, and Key players of your market. Present your Market Report & findings with an inbuilt presentation feature saving over 70% of your time and resources for Investor, Sales & Marketing, R&D, and Product Development pitches. VMI enables data delivery In Excel and Interactive PDF formats with over 15+ Key Market Indicators for your market.
About Us
Verified Market Research® stands at the forefront as a global leader in Research and Consulting, offering unparalleled analytical research solutions that empower organizations with the insights needed for critical business decisions. Celebrating 10+ years of service, VMR has been instrumental in providing founders and companies with precise, up-to-date research data.
With a team of 500+ Analysts and subject matter experts, VMR leverages internationally recognized research methodologies for data collection and analyses, covering over 15,000 high impact and niche markets. This robust team ensures data integrity and offers insights that are both informative and actionable, tailored to the strategic needs of businesses across various industries.
VMR's domain expertise is recognized across 14 key industries, including Semiconductor & Electronics, Healthcare & Pharmaceuticals, Energy, Technology, Automobiles, Defense, Mining, Manufacturing, Retail, and Agriculture & Food. In-depth market analysis cover over 52 countries, with advanced data collection methods and sophisticated research techniques being utilized. This approach allows for actionable insights to be furnished by seasoned analysts, equipping clients with the essential knowledge necessary for critical revenue decisions across these varied and vital industries.
Verified Market Research® is also a member of ESOMAR, an organization renowned for setting the benchmark in ethical and professional standards in market research. This affiliation highlights VMR's dedication to conducting research with integrity and reliability, ensuring that the insights offered are not only valuable but also ethically sourced and respected worldwide.
Follow Us On: LinkedIn | Twitter | Threads | Instagram | Facebook

Mr. Edwyne Fernandes Verified Market Research® US: +1 (650)-781-4080 US Toll Free: +1 (800)-782-1768 Email: sales@verifiedmarketresearch.com Web: https://www.verifiedmarketresearch.com/ SOURCE – Verified Market Research®

